ONWARD® Medical Announces Third Implant of BrainComputer Interface (BCI) System to Restore Movement after Spinal Cord Injury
ONWARD® Medical Announces Third Implant of BrainComputer Interface (BCI) System to Restore Movement after Spinal Cord Injury
Company continues pioneering research on BCI-enabled system to restore mobility after spinal cord injury
EINDHOVEN, the Netherlands — September 19, 2024 — ONWARD Medical N.V. (Euronext: ONWD), the medical technology company creating innovative spinal cord stimulation therapies to restore movement, function, and independence in people with spinal cord injury (SCI), announces another successful implant of its investigational ARC-BCI™ System to restore lower limb mobility after SCI.
The ARC-BCI System combines the investigational ONWARD ARC-IM® System (an implanted technology that delivers targeted stimulation to the spinal cord) with the investigational WIMAGINE® BCI from CEA-Clinatec to create a DigitalBridge™ across the injured spinal cord. The implant procedure was performed on September 12, 2024, by Jocelyne Bloch, MD, head of functional neurosurgery at Centre Hospitalier Universitaire Vaudois (CHUV) in Lausanne,
Switzerland.
“The procedure went smoothly, and early signs are encouraging,” said neurosurgeon Dr. Jocelyne Bloch. “We look forward to sharing more information in the coming months as the participant progresses in their rehabilitation and we publish observations from the study.”
This implant is part of an ongoing clinical feasibility study supported by a grant from the European Innovation Council under the Reverse Paralysis project and coordinated by .NeuroRestore, a Swiss neuroscience research institute. The researchers are also exploring use of the ONWARD ARC-BCI System to address upper limb movement challenges after SCI in a separate early feasibility clinical study funded by the Christopher & Dana Reeve Foundation.
The WIMAGINE BCI from CEA-Clinatec is an epidurally-implanted device with 7 years of human safety data; ONWARD ARC-IM Therapy has now been applied in more than 30 study participants.
“While other companies race to develop BCIs to communicate with and control computers, ONWARD Medical stands alone in our commitment to exploring the potential for this promising technology to restore movement of the human body after paralysis,” said Dave Marver, CEO of ONWARD Medical. “We salute our brilliant partners at CEA-Clinatec and .NeuroRestore for their important contributions to this research.”
This latest news marks the third human implant of the WIMAGINE BCI paired with ARC-IM Therapy to restore thought-enabled movement after SCI and the second for lower limb mobility. The first human use of ARC-BCI Therapy for lower limb mobility occurred in 2021, with results describing the individual’s augmented control over when and how he moved his paralyzed legs published in Nature in May 2023. In the fall of 2023, an individual was implanted to explore the potential for ARC-BCI Therapy to restore upper extremity function after SCI.
Earlier this year, the Company announced that it has been accepted into the US FDA’s new Total Product Lifecycle Advisory Program (TAP) for its ARC-BCI platform. Prior to the acceptance, the Company announced its ARC-BCI System was awarded FDA Breakthrough Device Designation (BDD), a requirement for TAP consideration. This is the Company’s 10th such award.
To learn more about ONWARD Medical’s commitment to partnering with the SCI Community to develop innovative solutions for restoring movement, function, and independence after spinal cord injury, please visit ONWD.com.
*All ONWARD® Medical devices and therapies, including but not limited to ARC-IM®, ARC-EX®, ARCBCI™, and ARC Therapy™, alone or in combination with a brain-computer interface (BCI), are investigational and not available for commercial use.
About ONWARD Medical
ONWARD® Medical is a medical technology company creating therapies to restore movement, function, and independence in people with spinal cord injury (SCI) and movement disabilities. Building on more than a decade of scientific discovery, preclinical, and clinical research conducted at leading hospitals, rehabilitation clinics, and neuroscience laboratories, the Company has developed ARC Therapy™, which has been awarded ten Breakthrough Device Designations from the US Food and Drug Administration (FDA).
ONWARD ARC Therapy is targeted, programmed spinal cord stimulation designed to be delivered by the Company’s external ARC-EX® or implantable ARC-IM® platforms. ARC Therapy can also be delivered by the Company’s ARC-BCI™ platform, which pairs the ARC-IM System with brain-computer interface (BCI) technology to restore movement after SCI with thought-driven control.
Use of non-invasive ARC-EX Therapy significantly improved upper limb function after SCI in the global pivotal Up-LIFT trial, with results published by Nature Medicine in May 2024. The Company has submitted its regulatory application to the FDA for clearance of the ARC-EX System in the US and is preparing for
regulatory submission in Europe. In parallel, the Company is conducting clinical studies with its ARC-IM Therapy, which demonstrated positive interim clinical outcomes for improved blood pressure regulation following SCI. Other ongoing clinical studies focus on using ARC-IM Therapy to address mobility after SCI and gait challenges in Parkinson’s disease as well as using the ARC-BCI platform to restore thoughtdriven movement of both upper and lower limbs after SCI.
Headquartered in Eindhoven, the Netherlands, ONWARD Medical has a Science and Engineering Center in Lausanne, Switzerland and a US office in Boston, Massachusetts. The Company is listed on Euronext Brussels and Amsterdam (ticker: ONWD).
For more information, visit ONWD.com, and connect with us on LinkedIn and YouTube.
For Media Inquiries:
Aditi Roy, VP Communications
For Investor Inquiries:
Amori Fraser, Finance Director
Disclaimer
Certain statements, beliefs, and opinions in this press release are forward-looking, which reflect the Company’s or, as appropriate, the Company directors’ current expectations and projections about future events. By their nature, forward-looking statements involve several risks, uncertainties, and assumptions that could cause actual results or events to differ materially from those expressed or implied by the forward-looking statements. These risks, uncertainties, and assumptions could adversely affect the outcome and financial effects of the plans and events described herein. A multitude of factors including,
but not limited to, delays in regulatory approvals, changes in demand, competition, and technology, can cause actual events, performance, or results to differ significantly from any anticipated development. Forward-looking statements contained in this press release regarding past trends or activities should not be taken as a representation that such trends or activities will continue in the future. As a result, the Company expressly disclaims any obligation or undertaking to release any update or revisions to any forward-looking statements in this press release as a result of any change in expectations or any change in events, conditions, assumptions, or circumstances on which these forward-looking statements are based. Neither the Company nor its advisers or representatives nor any of its subsidiary undertakings or any such person’s officers or employees guarantees that the assumptions underlying such forwardlooking statements are free from errors nor does either accept any responsibility for the future accuracy of the forward-looking statements contained in this press release or the actual occurrence of the forecasted developments. You should not place undue reliance on forward-looking statements, which speak only as of the date of this press release. All ONWARD Medical devices and therapies referenced here, including but not limited to ARC-IM®, ARC-EX®, ARC-BCI™ and ARC Therapy™, are investigational and not available for commercial use.
Carisma and Moderna Expand Collaboration to Develop Two In Vivo CAR-M Therapies for Autoimmune Diseases
Carisma and Moderna Expand Collaboration to Develop Two In Vivo CAR-M Therapies for Autoimmune Diseases
Building on successful pre-clinical in vivo CAR-M data in oncology, the companies will develop in vivo CAR-M for autoimmune diseases
Moderna nominated two autoimmune disease targets under the collaboration
Carisma is eligible to receive milestones and royalty payments
PHILADELPHIA, Sept. 10, 2024 /PRNewswire/ — Carisma Therapeutics Inc. (Nasdaq: CARM) (“Carisma” or the “Company”), a clinical stage biopharmaceutical company focused on discovering and developing innovative immunotherapies, today announced the expansion of its in vivo chimeric antigen receptor macrophage and monocyte (together, “CAR-M”) collaboration with Moderna, Inc. (Nasdaq: MRNA) to include the nomination of two targets for the treatment of autoimmune diseases. Carisma retains all rights in autoimmune disease beyond the two nominated targets, which will be exclusively partnered with Moderna.
Under this expanded collaboration, Carisma and Moderna will leverage Carisma’s proprietary CAR-M technology and Moderna’s mRNA/LNP platform to develop novel in vivo macrophage engineering approaches in the nominated autoimmune disease targets. Carisma will receive research funding and is eligible to receive development, regulatory, and commercial milestone payments, plus royalties on net sales of any products that are commercialized under the collaboration agreement. Carisma will be responsible for the discovery and optimization of development candidates, while Moderna will lead the clinical development and commercialization of therapeutics resulting from the agreement.
“We are excited to expand our collaboration with Moderna into the realm of autoimmune diseases,” said Steven Kelly, President and Chief Executive Officer of Carisma. “The nomination of the two autoimmune targets is a significant milestone in our mission to harness the power of macrophages to treat a broader range of diseases. Our innovative CAR-M technology has the potential to revolutionize the treatment landscape for patients suffering from these debilitating conditions.”
“We are excited to build on the progress of advancing in vivo CAR-M therapies with Carisma by expanding beyond oncology,” said Lin Guey, PhD, CSO of Therapeutic Research Ventures, Moderna. “We continue to believe that the combination of our platform and Carisma’s deep myeloid biology expertise could lead to innovative treatments for patients.”
The expanded collaboration between Carisma and Moderna underscores the potential of CAR-M technology to impact a diverse range of disease areas. The expansion will aim to bring transformative therapies to patients with cancer and autoimmune diseases, advancing the frontier of immunotherapy.
About Carisma Therapeutics
Carisma Therapeutics Inc. is a clinical stage biopharmaceutical company focused on utilizing our proprietary macrophage and monocyte cell engineering platform to develop transformative immunotherapies to treat cancer and other serious diseases. We have created a comprehensive, differentiated proprietary cell therapy platform focused on engineered macrophages and monocytes, cells that play a crucial role in both the innate and adaptive immune response. Carisma is headquartered in Philadelphia, PA. For more information, please visit www.carismatx.com.
Cautionary Note on Forward-Looking Statements
Statements in this press release about future expectations, plans and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, statements relating to Carisma’s business, strategy, future operations, cash runway, the advancement of Carisma’s product candidates and product pipeline, and clinical development of Carisma’s product candidates, including expectations regarding timing of initiation and results of clinical trials. The words “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “goals,” “intend,” “may,” “might,” “outlook,” “plan,” “project,” “potential,” “predict,” “target,” “possible,” “will,” “would,” “could,” “should,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
Any forward-looking statements are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in, or implied by, such forward-looking statements. These risks and uncertainties include, but are not limited to, (i) Carisma’s ability to realize the anticipated benefits of its pipeline reprioritization and corporate restructuring, (ii) Carisma’s ability to obtain, maintain and protect its intellectual property rights related to its product candidates; (iii) Carisma’s ability to advance the development of its product candidates under the timelines it anticipates in planned and future clinical trials and with its current financial and human resources; (iv) Carisma’s ability to replicate in later clinical trials positive results found in preclinical studies and early-stage clinical trials of its product candidates; (v) Carisma’s ability to realize the anticipated benefits of its research and development programs, strategic partnerships, research and licensing programs and academic and other collaborations; (vi) regulatory requirements or developments and Carisma’s ability to obtain and maintain necessary approvals from the U.S. Food and Drug Administration and other regulatory authorities related to its product candidates; (vii) changes to clinical trial designs and regulatory pathways; (viii) risks associated with Carisma’s ability to manage expenses; (ix) changes in capital resource requirements; (x) risks related to the inability of Carisma to obtain sufficient additional capital to continue to advance its product candidates and its preclinical programs; and (xi) legislative, regulatory, political and economic developments.
For a discussion of these risks and uncertainties, and other important factors, any of which could cause Carisma’s actual results to differ from those contained in the forward-looking statements, see the “Risk Factors” set forth in the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, its Quarterly Report on Form 10-Q for the quarter ended June 30, 2024, as well as discussions of potential risks, uncertainties, and other important factors in Carisma’s other recent filings with the Securities and Exchange Commission. Any forward-looking statements that are made in this press release speak as of the date of this press release. Carisma undertakes no obligation to revise the forward-looking statements or to update them to reflect events or circumstances occurring after the date of this press release, whether as a result of new information, future developments or otherwise, except as required by the federal securities laws.
Investors:
Shveta Dighe
Head of Investor Relations
Media Contact:
Julia Stern
(763) 350-5223
SOURCE Carisma Therapeutics Inc.

CorFlow Therapeutics AG completes €44 million Series B financing to support the advancement of a novel diagnostic and drug delivery platform for microvascular obstruction (MVO) in heart attack patients
CorFlow Therapeutics AG completes €44 million Series B financing to support the advancement of a novel diagnostic and drug delivery platform for microvascular obstruction (MVO) in heart attack patients
The Series B funding enables both an International Pivotal trial to diagnose MVO and a randomized controlled trial to evaluate therapeutic treatments for MVO through the CoFI system
BAAR, Switzerland–(BUSINESS WIRE)– CorFlow Therapeutics AG (CorFlow) announced today that it has raised €44 million in Series B funding, co-led by Broadview Ventures and Panakes Partners with strong continued support from 415 Capital, CorFlow’s initial VC investor and largest shareholder. Merieux Equity Partners, Laerdal Million Lives Fund, Wellington Partners, M&L Investments, Unorthodox Ventures, KOFA Healthcare and Monte Carlo Capital participated in the multinational syndicate.
Concurrent with the financing, David Prim of Broadview Ventures, Barbara Castellano of Panakes Partners, Yoann Bonnamour of Merieux Equity Partners, and Rhiya Pau of Laerdal Million Lives Fund have joined the CorFlow Board of Directors.
The Series B will fund the MOCA II (MVO with CoFITM System Assessment II) pivotal study intended to gain US market clearance. The trial will run in the US and Europe and aims to validate CorFlow’s CoFl system to diagnose MVO in heart attack patients immediately following stent implantation. Additionally, it will fund an adaptive platform therapy study evaluating treatment effects of therapeutic agents delivered locally through the CoFl system on heart attack patients diagnosed with MVO.
MVO affects more than half of all patients who suffer an acute heart attack and is an independent predictor for heart failure and mortality. Currently not routinely diagnosed MVO remains largely untreated, leading to poor patient outcomes, and contributing to high health care costs associated with cardiovascular disease.
CorFlow’s CoFl system is being developed to provide timely, accurate and consistent detection of MVO while patients are still in the catheter laboratory (cath lab) immediately following reopening of the larger epicardial arteries with a stent. Uniquely, the technology has been designed to also enable localized delivery of therapeutics to the microvasculature upon MVO diagnosis. CoFl also seamlessly integrates into standard workflows utilizing existing guide catheter and wire access.
“We are thrilled to have closed on this significant round of financing, backed by a top-tier global syndicate of medical technology investors,” commented Paul Mead, President and Chief Executive Officer of CorFlow. “Recent data from over 70 patients in our MOCA I first in human trial and from our extensive preclinical program support our collective confidence that we can improve outcomes in patients who suffer heart attacks, specifically those patients whose microvascular disease goes undiagnosed and untreated today. There is overwhelming evidence now that microvascular conditions are a significant root cause of adverse outcomes in heart attack patients and other cardiovascular conditions. I am grateful that our new investors have the vision to see ‘where the puck is going’ in this rapidly emerging field.”
The MOCA II IDE trial is designed to confirm the CoFl system’s accuracy in diagnosing MVO in high-risk heart attack patients. Led by Principal Investigator Dr. Tim Henry of The Christ Hospital in Cincinnati, Ohio, the trial will recruit several hundred patients undergoing stent implantation due to ST-Elevation Myocardial Infarction (STEMI) and will compare CoFl’s proprietary dynamic diagnostic measurement of MVO to post-procedure Contrast Enhanced Cardiac Magnetic Resonance Imaging (CMRI), the current gold standard for detecting MVO. The MOCA II trial leverages the learnings from the company’s First-In-Human MOCA I trial conducted in Switzerland, Lativa and the UK.
“There is increasing awareness of the impact of MVO on patient outcomes following coronary revascularization. CorFlow has the potential to be the next significant breakthrough in treating coronary artery disease, and Broadview is delighted to be supporting this impressive team and technology,” states David Prim of Broadview Ventures.
“CorFlow’s breakthrough technology has been designed by clinicians for clinicians and we are excited to support CorFlow’s mission to generate robust clinical data in order to get this much needed therapy to the patient as quickly as possible.” adds Barbara Castellano of Panakes Partners. “We also are committed to supporting the emerging field of microvascular disease in general, and this oversubscribed funding round gives us options to apply the technology in new ways, and in new geographies, where patients can benefit.”
The funding will also support a novel adaptive platform therapy trial in Europe evaluating whether localized delivery of therapeutics to the microvasculature immediately following stent implantation can improve outcomes in patients diagnosed with MVO. Led by Dr. Giovanni Luigi De Maria (Oxford University Hospitals Trust, UK) and Professor Colin Berry (University of Glasgow and NHS Golden Jubilee National Hospital, UK), this European RCT will assess the effects of several therapeutic agents with both clinical and imaging measures up to six months.
“The improvement in outcomes for heart attack patients has stagnated over many years now and we believe that real-time diagnosis and targeted therapy for MVO has the potential to save the lives of countless patients in the future.” comments Frederik Groenewegen of 415 Capital. “We have long been believers in the CorFlow technology and team, and with the support of this first-class investor syndicate we now have the opportunity to collect the clinical data required to establish a new standard of heart attack care.”
About CorFlow Therapeutics: Headquartered in Baar, Switzerland, with offices in Italy, and founded in 2016 by Dr. Rob Schwartz, Dr. Martin Rothman and Jon Hoem. CorFlow aspires to be the leader in diagnostic and therapeutic solutions for restoring healthy microvascular blood flow anywhere in the human body where a critical need exists. Working in close partnership with scientists from the University of Bern, ETH Zurich and the University Hospital Zurich, in a collaboration funded by the Swiss Innovation Agency (Innosuisse), CorFlow continues to explore applications in and beyond the heart.
About Microvascular Obstruction (MVO): Often described as the “last frontier” in the treatment of acute heart attacks, MVO is characterized by blockages in the microvascular coronary arteries, which vary in size down to the circumference of a human hair. Previous research has identified that MVO is one of the most powerful prognostic indicators for future adverse outcomes – for every 1% increase in MVO, there’s a corresponding 14% increase in one-year mortality risk and 8% increase in hospitalization due to heart failure.
Media and Scientific Contact
CorFlow Therapeutics AG

UroMems Raises Record $47 million (€44 Million) in Series C Financing to Fund Pivotal Clinical Trials of the UroActive™ System, the First Smart Automated Implant to Treat Stress Urinary Incontinence
UroMems Raises Record $47 million (€44 Million) in Series C Financing to Fund Pivotal Clinical Trials of the UroActive™ System, the First Smart Automated Implant to Treat Stress Urinary Incontinence
GRENOBLE, France & MINNEAPOLIS, Minnesota (June 25, 2024) – UroMems, a global company developing innovative, implantable mechatronics technology to treat stress urinary incontinence (SUI), today announced it has raised $47 million in its Series C financing, the largest fundraising round to date for the company. The capital will support large-scale U.S. and European pivotal clinical trials of the UroActive™ implant, paving the way for regulatory submission in multiple countries. UroActive is the first smart automated artificial urinary sphincter (AUS) to treat SUI.
The financing round was led by Crédit Mutuel Innovation and joined by the European Innovation Council as well as the existing investors Wellington Partners, Bpifrance, Supernova Invest, Hil-Invent, b-to-v Partners and Financière Arbevel.
“We are thrilled to lead this record round for UroMems,” said Jérôme Feraud, Head of Health Division at Crédit Mutuel Innovation. “Based on very positive data and feedback from patients and physicians in the pilot clinical trials, we strongly believe that the UroActive smart implant has the potential to become the standard of care as the next-generation SUI treatment.”
Under the strong and steady leadership, energy and guidance of co-founders Chief Executive Officer Hamid Lamraoui and Chief Medical Officer Professor Pierre Mozer, UroMems has successfully surpassed critical milestones in research and development, clinical outcomes and building the organization. The funding comes on the heels of exceptionally strong results from the first-in-man multicenter clinical study six-month endpoint, and successful six-month primary endpoint for the first-ever female patient to receive the UroActive System. For all study patients the devices are operating as expected for over one year now with no need for revision nor explant. In addition, extremely positive follow-up was received on secondary outcomes measures, including leak rate values and patient quality of life questionnaires.
“We’ve invested in UroMems from the start and our confidence that the UroActive System will improve patients’ lives continues to grow as the company reaches one major milestone after the other,” said Dr. Johannes Fischer, Partner at Wellington Partners. “We look forward to continue supporting the company as they move closer to bringing this promising technology to both women and men suffering from SUI.”
“On behalf of our entire exceptional team and the patients whose lives have been restored thanks to our technology, we are grateful to our investor partners for their continued support as we approach the critical milestone of launching our pivotal clinical trial,” said Lamraoui. “The confidence of our new and existing investors combined with the strength of our compelling results and tremendous physician interest shows the high potential of our technology. This brings us one step closer to delivering on the massive unmet need for men, women and physicians desperately seeking a better SUI treatment option.”
SUI, or involuntary urinary leakage, affects an estimated 40 million Americans and 90 million Europeans. SUI significantly impacts quality of life, as it can be debilitating, and often leads to depression, low selfesteem and social stigma.
About UroActive
UroActive is an active implantable electronic artificial urinary sphincter that is being developed to compensate for sphincter insufficiency in patients, both men and women, with SUI. It is based on a unique bionic platform using embedded smart, digital and robotic systems which, based on data collected from a patient, create a treatment algorithm that is specific for each patient’s needs. The UroMems technology platform is protected by more than 150 patents and is designed to overcome the limitations of current solutions by optimizing safety and performance, patient experience and surgeonconvenience. UroActive has not received marketing authorization from the FDA and is not available for sale in the United States or in the EU.
For more information, please visit www.uromems.com.
Media Contact:
Shelli Lissick
651-276-6922

Carisma Therapeutics Granted FDA Fast Track Designation for CT-0525 for the Treatment of HER2-overexpressing Solid Tumors
Carisma Therapeutics Granted FDA Fast Track Designation for CT-0525 for the Treatment of HER2-overexpressing Solid Tumors
Fast Track designation highlights potential for CT-0525 to address unmet need for patients with solid tumors
Initial Phase 1 data expected by year-end 2024
PHILADELPHIA, June 25, 2024 /PRNewswire/ — Carisma Therapeutics Inc. (Nasdaq: CARM) (“Carisma” or the “Company”), a clinical stage biopharmaceutical company focused on discovering and developing innovative immunotherapies, announced today that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation for CT-0525, an ex vivo gene-modified autologous chimeric antigen receptor-monocyte (CAR-Monocyte) cellular therapy intended to treat solid tumors that overexpress human epidermal growth factor receptor 2 (HER2).
The FDA’s Fast Track program is aimed to facilitate the development, and expedite the review, of novel potential therapies that are designed to treat serious conditions and have the potential to address significant unmet medical need.
“Receiving Fast Track designation for CT-0525 from the FDA marks a significant milestone for Carisma, highlighting the FDA’s recognition of the serious and life-threatening nature of these malignancies and the potential of CT-0525 to meet this critical medical need,” said Eugene P. Kennedy, M.D., Chief Medical Officer of Carisma. “We are committed to working closely with the FDA to accelerate the development of CT-0525. Currently, we are enrolling patients in the Phase 1 clinical trial and remain on track to report initial clinical data by the end of 2024.”
The Phase 1 clinical trial for CT-0525 is an open-label study designed to assess the safety, tolerability, and manufacturing feasibility of CT-0525. This trial will enroll participants with locally advanced (unresectable) or metastatic solid tumors overexpressing HER2 whose disease has progressed on standard approved therapies. The initial study design will consist of two dose escalation cohorts. Further details of the trial can be found at www.clinicaltrials.gov under NCT identifier: NCT06254807.
About CT-0525
CT-0525 is a first-in-class, ex vivo gene-modified autologous chimeric antigen receptor-monocyte (CAR-Monocyte) cellular therapy intended to treat solid tumors that overexpress human epidermal growth factor receptor 2 (HER2). It is being studied in a multi-center, open label, Phase 1 clinical trial for patients with advanced/metastatic HER2-overexpressing solid tumors that have progressed on available therapies. The CAR-Monocyte approach has the potential to address some of the challenges of treating solid tumors with cell therapies, including tumor infiltration, immunosuppression within the tumor microenvironment, and antigen heterogeneity. CT-0525 has the potential to enable significant dose escalation, enhance tumor infiltration, increase persistence, and reduce manufacturing time compared to macrophage therapy.
About Carisma Therapeutics
Carisma Therapeutics Inc. is a clinical stage biopharmaceutical company focused on utilizing our proprietary macrophage and monocyte cell engineering platform to develop transformative immunotherapies to treat cancer and other serious diseases. We have created a comprehensive, differentiated proprietary cell therapy platform focused on engineered macrophages and monocytes, cells that play a crucial role in both the innate and adaptive immune response. Carisma is headquartered in Philadelphia, PA. For more information, please visit www.carismatx.com.
Cautionary Note on Forward-Looking Statements
Statements in this press release about future expectations, plans and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, statements relating to Carisma’s business, strategy, future operations, cash runway, the advancement of Carisma’s product candidates and product pipeline, and clinical development of Carisma’s product candidates, including expectations regarding timing of initiation and results of clinical trials. The words “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “goals,” “intend,” “may,” “might,” “outlook,” “plan,” “project,” “potential,” “predict,” “target,” “possible,” “will,” “would,” “could,” “should,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
Any forward-looking statements are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in, or implied by, such forward-looking statements. These risks and uncertainties include, but are not limited to, (i) Carisma’s ability to realize the anticipated benefits of its pipeline reprioritization and corporate restructuring, (ii) Carisma’s ability to obtain, maintain and protect its intellectual property rights related to its product candidates; (iii) Carisma’s ability to advance the development of its product candidates under the timelines it anticipates in planned and future clinical trials and with its current financial and human resources; (iv) Carisma’s ability to replicate in later clinical trials positive results found in preclinical studies and early-stage clinical trials of its product candidates; (v) Carisma’s ability to realize the anticipated benefits of its research and development programs, strategic partnerships, research and licensing programs and academic and other collaborations; (vi) regulatory requirements or developments and Carisma’s ability to obtain and maintain necessary approvals from the U.S. Food and Drug Administration and other regulatory authorities related to its product candidates; (vii) changes to clinical trial designs and regulatory pathways; (viii) risks associated with Carisma’s ability to manage expenses; (ix) changes in capital resource requirements; (x) risks related to the inability of Carisma to obtain sufficient additional capital to continue to advance its product candidates and its preclinical programs; and (xi) legislative, regulatory, political and economic developments.
For a discussion of these risks and uncertainties, and other important factors, any of which could cause Carisma’s actual results to differ from those contained in the forward-looking statements, see the “Risk Factors” set forth in the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, the Company’s Quarterly Report on Form 10-Q for the Quarter ended March 31, 2024, as well as discussions of potential risks, uncertainties, and other important factors in Carisma’s other recent filings with the Securities and Exchange Commission. Any forward-looking statements that are made in this press release speak as of the date of this press release. Carisma undertakes no obligation to revise the forward-looking statements or to update them to reflect events or circumstances occurring after the date of this press release, whether as a result of new information, future developments or otherwise, except as required by the federal securities laws.
Investors:
Shveta Dighe
Head of Investor Relations
Media Contact:
Julia Stern
(763) 350-5223

Carisma Therapeutics Presents Preclinical Proof of Concept Data Demonstrating the Anti-Fibrotic Potential of Engineered Macrophages at ASGCT 2024
Carisma Therapeutics Presents Preclinical Proof of Concept Data Demonstrating the Anti-Fibrotic Potential of Engineered Macrophages at ASGCT 2024
Novel anti-fibrotic engineered macrophage therapy reduced liver fibrosis in preclinical models
Development candidate nomination expected in the first quarter of 2025
“We are pleased to unveil preclinical proof of concept data for our liver fibrosis program, which highlight the potential of engineered macrophages to combat a prevalent disease that is associated with late-stage metabolic dysfunction-associated steatohepatitis (MASH) and represents a significant unmet need,” said Michael Klichinsky, PharmD, PhD, Co-founder and Chief Scientific Officer of Carisma. “The data, from two independent models, demonstrate that engineered macrophages trafficked to fibrotic tissues, expressed genetically encoded disease-modifying payloads, and significantly reduced fibrosis in the liver. Given these encouraging data, we look forward to further progressing the liver fibrosis program, which is our first expansion outside of oncology.”
In the presentation titled “Genetically Engineered Macrophage Cell Therapy Reverses Liver and Lung Fibrosis in Preclinical Models,” Carisma presented preclinical proof-of-concept data for engineered macrophage cell therapy in liver fibrosis. In liver models, the data showed that a single dose of macrophages co-expressing the anti-fibrotic factor relaxin and the anti-inflammatory cytokine IL10 significantly improved established fibrosis in a CCl4-induced liver fibrosis model, with a 116% reduction in fibrosis relative to untreated control. Also, systemic administration of engineered macrophages co-expressing relaxin and IL10 significantly reduced liver fibrosis in a high fat diet MASH model, with a 45% reduction in fibrosis relative to untreated control. In both models, the relaxin-IL10 macrophage treatment also resulted in a greater reduction in liver fibrosis compared to non-engineered macrophages.
The presentation also included initial data for the use of engineered macrophages in pulmonary fibrosis. The data showed that a single dose of macrophages expressing a dominant negative TGFβ receptor, which nullified pro-fibrotic TGFβ signaling in the lung, prevented fibrosis in a bleomycin mouse model of pulmonary fibrosis, with a 90% reduction in fibrosis relative to untreated control.
Carisma expects to nominate a development candidate for its liver fibrosis program in the first quarter of 2025.
The poster presented at ASGCT 2024 is now available online in the “Publications” section of Carisma’s website at https://carismatx.com/technology/publications/
About Carisma
Carisma Therapeutics Inc. is a clinical stage biopharmaceutical company focused on utilizing our proprietary macrophage and monocyte cell engineering platform to develop transformative immunotherapies to treat cancer and other serious diseases. We have created a comprehensive, differentiated proprietary cell therapy platform focused on engineered macrophages and monocytes, cells that play a crucial role in both the innate and adaptive immune response. Carisma is headquartered in Philadelphia, PA. For more information, please visit www.carismatx.com.
Cautionary Note on Forward-Looking Statements
Statements in this press release about future expectations, plans and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, statements relating to Carisma’s business, strategy, future operations, cash runway, the advancement of Carisma’s product candidates and product pipeline, and clinical development of Carisma’s product candidates, including expectations regarding timing of initiation and results of clinical trials. The words “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “goals,” “intend,” “may,” “might,” “outlook,” “plan,” “project,” “potential,” “predict,” “target,” “possible,” “will,” “would,” “could,” “should,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
Any forward-looking statements are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in, or implied by, such forward-looking statements. These risks and uncertainties include, but are not limited to, (i) Carisma’s ability to obtain, maintain and protect its intellectual property rights related to its product candidates; (ii) Carisma’s ability to advance the development of its product candidates under the timelines it anticipates in planned and future clinical trials and with its current financial and human resources; (iii) Carisma’s ability to replicate in later clinical trials positive results found in preclinical studies and early-stage clinical trials of its product candidates; (iv) Carisma’s ability to realize the anticipated benefits of its research and development programs, strategic partnerships, research and licensing programs and academic and other collaborations; (v) regulatory requirements or developments and Carisma’s ability to obtain and maintain necessary approvals from the U.S. Food and Drug Administration and other regulatory authorities related to its product candidates; (vi) changes to clinical trial designs and regulatory pathways; (vii) risks associated with Carisma’s ability to manage expenses; (viii) changes in capital resource requirements; (ix) risks related to the inability of Carisma to obtain sufficient additional capital to continue to advance its product candidates and its preclinical programs; and (x) legislative, regulatory, political and economic developments.
For a discussion of these risks and uncertainties, and other important factors, any of which could cause Carisma’s actual results to differ from those contained in the forward-looking statements, see the “Risk Factors” set forth in the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, as well as discussions of potential risks, uncertainties, and other important factors in Carisma’s other recent filings with the Securities and Exchange Commission. Any forward-looking statements that are made in this press release speak as of the date of this press release. Carisma undertakes no obligation to revise the forward-looking statements or to update them to reflect events or circumstances occurring after the date of this press release, whether as a result of new information, future developments or otherwise, except as required by the federal securities laws.
Investors:
Shveta Dighe
Head of Investor Relations
Media Contact:
Julia Stern
(763) 350-5223

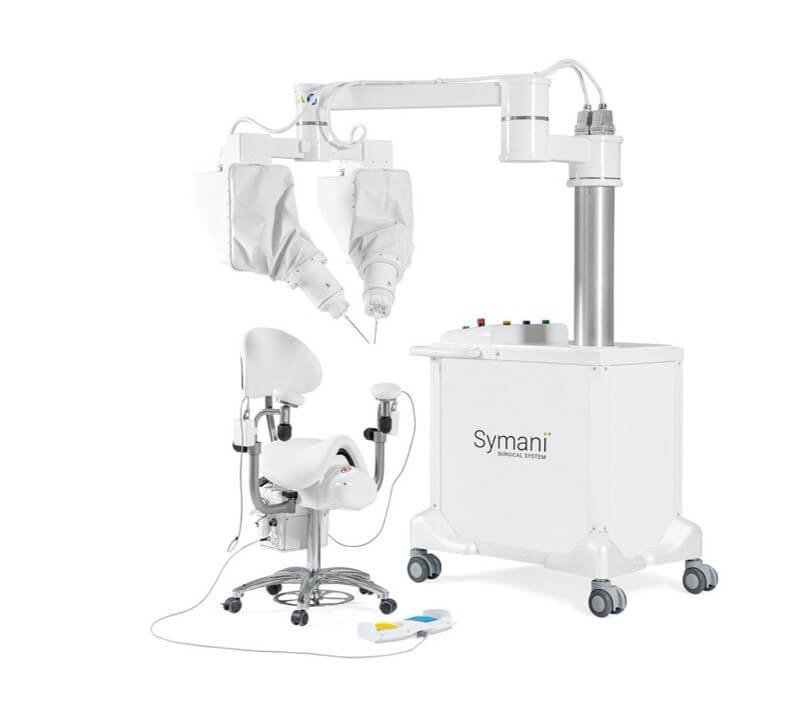
MMI Receives FDA Authorization to Commercialize Symani® Surgical System in the U.S.
MMI Receives FDA Authorization to Commercialize Symani® Surgical System in the U.S.
De Novo Classification builds on proven clinical success and expands global access to new category of treatments
JACKSONVILLE, Fla. – April 8, 2024 – MMI (Medical Microinstruments, Inc.), a robotics company dedicated to increasing treatment options and improving clinical outcomes for patients with complex conditions, today announced that its Symani Surgical System is now commercially available in the United States. The U.S. Food and Drug Administration (FDA) granted De Novo Classification to the robotic system for soft tissue manipulation to perform microsurgery, a highly specialized technique that involves reconnecting tiny vessels to restore blood flow or redirect fluid during reconstruction or repair.
The FDA authorization makes the Symani Surgical System the only commercially available platform in the U.S. for reconstructive microsurgery. The technology is positioned to open the field of microsurgery to new surgeons by quickly developing their skills, as well as to empower skilled microsurgeons to confidently expand into supermicrosurgery, creating a novel category of treatments that the human hand cannot perform without robotic assistance.
“The U.S. is facing a potentially dire shortage of physicians, and that shortage acutely impacts specialized fields of medicine, such as microsurgery,” said Mark Toland, CEO of MMI. “With the authorization from the FDA, our technology will expand its reach to pioneering hospitals in the U.S. It will help those hospitals grow their open surgical programs, expand the number of physicians who can perform these highly complicated procedures, and increase patient access to the most advanced techniques for surgeries in complex disease states, such as lymphedema. Our system will continue to provoke surgeons to challenge their definitions of ‘treatable’ and ‘untreatable’ and empower them to solve cases that have historically been too difficult to treat.”
Specific techniques used in microsurgery include reconnecting small anatomical structures, such as blood and lymphatic vessels, during open surgical procedures. Supermicrosurgery involves reconstruction or repair of even smaller vessels, typically less than 1mm in diameter, and fewer than 600 surgeons worldwide perform supermicrosurgery today.
The Symani Surgical System provides advanced solutions for a range of open surgeries, including post-mastectomy breast cancer reconstruction, extremity reconstruction using free tissue transfer, and lymphatic system repair.
“By making open surgery less invasive and more precise, we can treat more conditions and offer robotic-assisted surgical options to patients that simply do not exist today,” said Dr. L. Scott Levin, co-CMO of MMI. “Within the next five years, this expanded portfolio of addressable open surgical procedures is expected to exceed the number of eligible laparoscopic, or minimally invasive, procedures that leverage robotic assistance. The authorization from the FDA helps to solve a critical unmet need and will help surgeons perform a new category of complex open surgeries enabled by transformative technology.”
The Symani Surgical System offers surgeons entirely new capabilities because it features the world’s smallest surgical robotic wrist, called NanoWrist®. The unique design enables surgeons to replicate the natural movements of the human hand at the micro scale, which encourages a flatter learning curve in the training process. The articulated wrist features seven degrees of freedom that match the human wrist, tremor filtration and motion scaling, ultimately increasing precision and control.
Surgeons have leveraged the Symani Surgical System in nearly 1,000 clinical cases in the European Union and in thousands of preclinical cases around the world. The Symani Surgical System is available for commercial use in Europe and parts of Asia Pacific. MMI plans to immediately launch the technology in the U.S.
To learn more about MMI and the Symani Surgical System, visit MMI’s website here: https://mmimicro.com.
MMI (Medical Microinstruments, Inc.) is on a mission to advance robotic technology that pushes the limits of soft tissue open surgery and opens new opportunities for surgeons to restore quality of life for more patients with complex conditions. The company was founded in 2015 near Pisa, Italy, and its proprietary Symani® Surgical System combines the world’s smallest wristed microinstruments with tremor-reducing and motion-scaling technologies to address significant unmet patient needs across the globe. This first-of-its-kind surgical robotic platform for open, soft tissue micro-level surgery can help address microvascular repair and lymphatic repair. In Europe, it also addresses peripheral nerve repair. The Symani System is authorized for use in the U.S. by the FDA and is a CE Marked medical device in Europe. MMI is backed by global investors including Fidelity Management & Research Company, Andera Partners, BioStar, Deerfield Management, Fountain Healthcare Partners, Panakès Partners, RA Capital, Sambatech, and Wellington Partners.

ONWARD® Medical Submits De Novo Application to FDA for its ARC-EX® System
THIS PRESS RELEASE CONTAINS INSIDE INFORMATION WITHIN THE MEANING OF ARTICLE 7(1) OF THE EUROPEAN MARKET ABUSE REGULATION (596/2014)
ONWARD® Medical Submits De Novo Application to FDA for its ARC-EX® System
FDA clearance would allow the Company to market its breakthrough therapy to improve or restore hand and arm function after spinal cord injury in the US
EINDHOVEN, the Netherlands — April 2, 2024 — ONWARD Medical N.V. (Euronext: ONWD), the medical technology company creating innovative spinal cord stimulation therapies to improve or restore movement, function, and independence in people with spinal cord injury (SCI), today announces it has submitted its De Novo application to the US Food and Drug Administration (FDA) to allow marketing of its breakthrough ARC-EX System to restore function of the upper extremities after SCI.
The submission marks an historic milestone for the Company in its mission to restore mobility and function for people with SCI. Once cleared by the FDA, ARC-EX will be the first-ever spinal cord stimulation therapy to restore hand and arm function after SCI and the first commercial product for ONWARD Medical. ONWARD prioritized upper limb function as its first indication for the ARC-EX System given feedback from the SCI Community of the importance of arm, hand, and finger function in empowering independence after SCI.
“We are delighted to be one step closer to bringing our breakthrough ARC-EX System to people living with SCI after submitting this De Novo application for regulatory clearance in the United States,” said ONWARD Medical CEO Dave Marver. “This therapy has the potential to transform the lives of people living with paralysis, while also positively impacting their loved ones.”
The De Novo application follows the Company’s global pivotal study – called Up-LIFT – the first large-scale pivotal study of transcutaneous spinal cord stimulation. The study investigated the safety and effectiveness of ARC-EX Therapy in improving upper limb strength and function in 65 study participants with chronic tetraplegia at 14 leading SCI neurorehabilitation centers in the United States, Canada, the United Kingdom, and the Netherlands. The study met all primary safety and effectiveness endpoints and demonstrated that 72% of participants responded to ARC-EX Therapy1, showing improvement both in strength and function.
“The SCI Community is eager to have access to this innovative technology,” said Candy Tefertiller, PT, DPT, PhD, NCS, Executive Director of Research and Evaluation, Craig Hospital in Lakewood, Colorado. “Even a small difference in hand and arm function can have a profound impact on independence and quality of life. The results of the Up-LIFT trial that led to this submission represent a significant advancement in the use of neuromodulation for individuals with spinal cord injury.”
The ONWARD ARC-EX System delivers ARC-EX Therapy™ – targeted, programmed electrical stimulation – transcutaneously to the spinal cord to increase strength, movement, and function of the upper limbs after SCI. The ARC-EX System was previously awarded FDA Breakthrough Device Designation (BDD) for upper limb function, which provided prioritized FDA review, the opportunity to interact with FDA experts, and the potential for additional reimbursement.
Nearly 200,000 people in the US and Europe have incomplete impaired upper extremity function after spinal cord injury.2,3 The Company is preparing for regulatory submission in Europe next.
To learn more about ONWARD Medical’s commitment to partnering with the SCI Community to develop innovative solutions for restoring movement, function, and independence after spinal cord injury, please visit ONWD.com.
*All ONWARD Medical devices and therapies, including but not limited to ARC-IM®, ARC-EX®, ARCBCI™, and ARC Therapy™, alone or in combination with a brain-computer interface (BCI), are investigational and not available for commercial use.
About ONWARD Medical
ONWARD Medical is a medical technology company creating therapies to restore movement, function, and independence in people with spinal cord injury (SCI) and movement disabilities. Building on more than a decade of science and preclinical research conducted at leading neuroscience laboratories, the Company has received ten Breakthrough Device Designations from the US Food and Drug Administration for its ARC Therapy™ platform.
ONWARD® ARC Therapy, which can be delivered by external ARC-EX® or implantable ARC-IM® systems, is designed to deliver targeted, programmed spinal cord stimulation. Positive results were presented in 2023 from the Company’s pivotal study, called Up-LIFT, evaluating the ability for transcutaneous ARC Therapy to improve upper extremity strength and function. The Company has submitted its De Novo regulatory clearance submission for ARC-EX for the US and is preparing for regulatory submission in Europe. In parallel, the Company is conducting studies with its implantable ARC-IM platform, which
demonstrated positive interim clinical outcomes for improved blood pressure regulation, a component of hemodynamic instability, following SCI. Other ongoing studies include combination use of ARC-IM with a brain-computer interface (BCI) to address multiple symptoms of SCI.
Headquartered in Eindhoven, the Netherlands, ONWARD Medical has a Science and Engineering Center in Lausanne, Switzerland and a US office in Boston, Massachusetts. The Company also has an academic partnership with .NeuroRestore, a collaboration between the Swiss Federal Institute of Technology
(EPFL), and Lausanne University Hospital (CHUV).
ONWARD Medical is listed on Euronext Brussels and Amsterdam (ticker: ONWD).
For more information, visit ONWD.com, and connect with us on LinkedIn and YouTube.
For Media Enquiries:
Aditi Roy, VP Communications
For Investor Enquiries:
Khaled Bahi, Interim CFO
Disclaimer
Certain statements, beliefs, and opinions in this press release are forward-looking, which reflect the Company’s or, as appropriate, the Company directors’ current expectations and projections about future events. By their nature, forward-looking statements involve several risks, uncertainties, and assumptions
that could cause actual results or events to differ materially from those expressed or implied by the forward-looking statements. These risks, uncertainties, and assumptions could adversely affect the outcome and financial effects of the plans and events described herein. A multitude of factors including, but not limited to, changes in demand, competition, and technology, can cause actual events, performance, or results to differ significantly from any anticipated development. Forward-looking statements contained in this press release regarding past trends or activities should not be taken as a representation that such trends or activities will continue in the future. As a result, the Company expressly disclaims any obligation or undertaking to release any update or revisions to any forward-looking statements in this press release as a result of any change in expectations or any change in events, conditions, assumptions, or circumstances on which these forward-looking statements are based. Neither the Company nor its advisers or representatives nor any of its subsidiary undertakings or any such person’s officers or employees guarantees that the assumptions underlying such forward-looking statements are free from errors nor does either accept any responsibility for the future accuracy of the forward-looking statements contained in this press release or the actual occurrence of the forecasted developments. You should not place undue reliance on forward-looking statements, which speak only as of the date of this press release. All ONWARD Medical devices and therapies referenced here, including but not limited to ARC-IM®, ARC-EX®, ARC-BCI™ and ARC Therapy™, are investigational and not available for commercial use.
Carisma Therapeutics Announces Changes to its Board of Directors
Carisma Therapeutics Announces Changes to its Board of Directors
Appointment of John Hohneker, M.D.
Resignation of Chidozie Ugwumba
PHILADELPHIA, April 1, 2024 /PRNewswire/ — Carisma Therapeutics Inc. (Nasdaq: CARM) (“Carisma” or the “Company”), a clinical-stage biopharmaceutical company focused on discovering and developing innovative immunotherapies, today announced the appointment of John Hohneker, M.D. to the Board of Directors of the Company, effective April 1, 2024. Additionally, the Company announced that Chidozie Ugwumba, who has served on Carisma’s Board of Directors since December 2020, has advised the Board of his intent to step down from his role as a member of the Board and chair of the Audit Committee of the Board effective April 1, 2024, as a result of his other professional commitments.
“We are pleased to welcome John to the Carisma Board of Directors,” said Sanford Zweifach, Chair of the Carisma Board of Directors. “I believe John’s extensive track record of clinical development, strategic leadership, and public company Board membership in the biopharmaceutical space will enable him to immediately support the Company as it continues leading the field of engineered macrophage development. We would also like to thank Chidozie for his invaluable advice and guidance since he first joined the Board over three years ago.”
“I am excited to join Carisma’s Board as the Company continues to develop innovative immunotherapies,” said Dr. Hohneker. “I am looking forward to partnering with the Board and the management team as they work to potentially bring novel treatments to patients in need across multiple therapeutic areas.”
Dr. Hohneker brings over 30 years of extensive experience in drug development and leadership across the biotech and pharmaceutical sectors. He most recently served as President and Chief Executive Officer of Anokion SA. Prior to this, he held the role of President of Research and Development at FORMA Therapeutics Inc. Before joining FORMA Therapeutics, Dr. Hohneker held roles of increasing responsibility at Novartis AG, including most recently as Senior Vice President and Global Head of Development, Immunology, and Dermatology. Prior to his time at Novartis, Dr. Hohneker held positions of increasing responsibility at Glaxo Wellcome and its legacy company, Burroughs Wellcome.
Dr. Hohneker currently serves on the Board of Directors of Curis, Inc., Sonata Therapeutics, Inc., and Trishula Therapeutics, Inc. Previously, Dr. Hohneker has served as a member of the Board of Directors of Torque Therapeutics, Inc., Dimension Therapeutics, Inc., Cygnal Therapeutics, Inc., BioTheryX Inc., Evelo Biosciences, Inc., Humanigen, Inc. and Aravive, Inc. Dr. Hohneker received his bachelor’s degree in chemistry from Gettysburg College and his M.D. from the Rutgers School of Biomedical and Health Sciences (formerly the University of Medicine and Dentistry of New Jersey-Rutgers Medical School). He completed his internship and residency in internal medicine and his fellowship in medical oncology, all at the University of North Carolina at Chapel Hill.
About Carisma
Carisma Therapeutics Inc. is a clinical-stage biopharmaceutical company focused on utilizing our proprietary macrophage and monocyte cell engineering platform to develop transformative immunotherapies to treat cancer and other serious diseases. We have created a comprehensive, differentiated proprietary cell therapy platform focused on engineered macrophages and monocytes, cells that play a crucial role in both the innate and adaptive immune response. Carisma is headquartered in Philadelphia, PA. For more information, please visit www.carismatx.com.
TRiCares Announces the Appointment of Ahmed Elmouelhi as President & Chief Executive Officer
Paris, France and Munich, Germany, March 25, 2024 – TRiCares SAS (“TRiCares”) a privately held pioneer in the field of minimally invasive treatment of tricuspid regurgitation, today is pleased to announce the appointment of Ahmed Elmouelhi as President & Chief Executive Officer (CEO). In conjunction with this, and following eight years at the helm of the business, Helmut Straubinger is retiring as President & CEO.
Ahmed Elmouelhi is a seasoned professional with more than 20 years of medical device experience across strategy, sales and marketing, R&D, and clinical science. Before joining TRiCares, Ahmed served as the Senior Vice President of Product Marketing, Strategy, and Business Development at AtriCure. There, he was instrumental in the establishment of Left Atrial Appendage (LAA) therapies and the creation of two successful business franchises for the company. Previously, Ahmed held roles of increasing responsibility at Medtronic, within its Structural Heart division, as well as AGA Medical (now part of Abbott Laboratories), where he led cross-functional teams to launch multiple generations of Transcatheter Aortic Valve Replacement (TAVR) therapies and accessories, Congenital Heart Defect therapies, and percutaneous LAA devices.
Ahmed studied at Massachusetts Institute of Technology where he obtained both his Masters and Bachelors in Mechanical Engineering with a minor in Economics. He currently serves as the Chairman of the American Heart Association in Minnesota.
Since joining the business in March 2016, Helmut has overseen the development of the first European minimally invasive implantable tricuspid valve, bringing it to first-in-man clinical use. Helmut will work with Ahmed to provide a seamless transition before his retirement at the end of March and will continue to support the team on a consultancy basis.
Eric Fain, MD, Chairman of TRiCares, commented: “I would like to extend my thanks to Helmut, on behalf of the board of directors, for his efforts over many years in leading TRiCares to successfully develop the leading transcatheter tricuspid replacement technology and demonstrate its initial strong clinical performance. After evaluating many candidates for the next CEO, I am confident that Ahmed is the right person to drive the company’s success as it enters its next phase.”
Ahmed Elmouelhi, newly appointed President & CEO of TRiCares, added: “This is an exciting time to be joining the business, and I am looking forward to leading the team over the coming months and years. The potential for Topaz is clear, and the need great, with 1.8 million patients suffering from Tricuspid heart valve regurgitation in the US alone. Helmut leaves a significant legacy after many years with the business, and I wish him the best for his retirement.”
Helmut Straubinger, former President & CEO of TRiCares, commented: “I would like to express my heartfelt gratitude to the entire TRiCares team for their unwavering commitment and hard work over the last eight years. As the company looks towards the US, this is the right moment for a change in leadership. I look forward to following TRiCares’ progress under Ahmed’s guidance, as it seeks to benefit Tricuspid regurgitation patients globally.”
Press inquiries: